SingHealth Duke-NUS Academic Medical Centre will NEVER ask you to transfer money over a call. If in doubt, call the 24/7 ScamShield helpline at 1799, or visit the ScamShield website at www.scamshield.gov.sg.
Landmark study launched to detect liver cancer early in Singapore
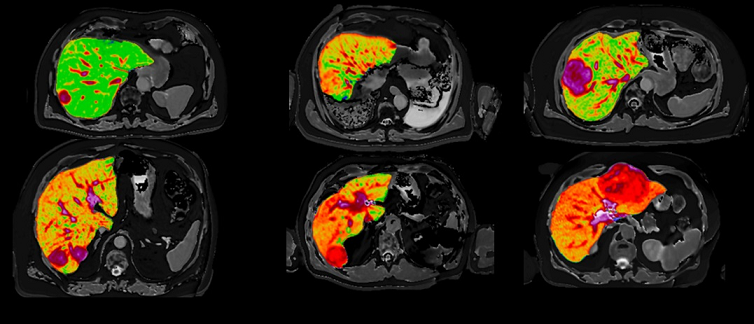
Quantitative MR images of the liver from six patients presenting with liver cancer produced by Perspectum’s LiverMultiScan which will be used in a new study led by National Cancer Centre Singapore. (Image credit: Mole DJ et al. Plos One. 2020;15(12):e0238568)
A first of its kind cohort study on patients at high risk of developing primary liver cancer also known as hepatocellular carcinoma (HCC), has been launched to diagnose HCC more accurately at an earlier stage and to predict an individual’s likelihood of developing the cancer. HCC is the sixth most common cancer in the world but the third most common cause of cancer deaths globally. [1] In Singapore, HCC is the third and fourth most common cause of cancer deaths, amongst males and females respectively. [2] While potentially curative treatment is possible with early diagnosis, only 20% of HCC cases are detected at a stage where cure is possible. This investigator-initiated multi-centre study led by the National Cancer Centre of Singapore (NCCS) called EarLy DEtection of HCC: miRNA, microbiome and imaGing biomArkers in the evolution of chroNiC livEr Disease in a high-risk prospective cohort (ELEGANCE), addresses this urgent, unmet need for individuals at high risk of developing HCC.
While individuals at high risk of developing HCC are well defined clinically as those with pre-existing liver cirrhosis, chronic viral hepatitis and/or fatty liver, there are currently no validated diagnostic, predictive and prognostic biomarkers for HCC, making early diagnosis challenging. Identifying such biomarkers would greatly improve patient outcomes. Currently, emerging data suggest that the evolution of chronic liver disease into HCC may be reflected by changes in the stool (microbiome), blood and urine (metabolome). An AI algorithm that leverages MRI imaging parameters may also be able to predict an individual’s risk of developing HCC by offering personalised prediction.
The ELEGANCE study will enrol patients at high-risk of developing HCC to deliver robust scientific data with the aim of developing more precise clinical tools to diagnose HCC at an early stage and predict which individuals are at highest risk. In addition, the study will highlight how the microbiome and metabolome changes with disease development and identify potential therapeutic targets that may slow disease progression and reduce the risks of developing cancer.
“Having cared for patients with liver cancer for more than 20 years, I see a pressing need to better predict this disease, diagnose it early and improve treatment outcomes,” said Prof Pierce Chow, Principal Investigator of the study and Senior Consultant, Division of Surgery and Surgical Oncology, Singapore General Hospital (SGH) and NCCS. “The ELEGANCE study is the first in the world to prospectively investigate the role of microRNA (miRNA), microbiome, metabolome and imaging biomarkers in the evolution of chronic liver disease and the early diagnosis of liver cancer.”
“This landmark study is designed to provide in-depth and multi-faceted data on the prognosis of HCC which can offer important insights to developing potential new diagnostic and predictive tools for this cancer,” said Assistant Professor Mihir Gandhi, Health Services & Systems Research Signature Programme and Head of Biostatistics Core team at the Centre for Quantitative Medicine, Duke-NUS Medical School (Duke-NUS), Singapore, whose team shaped the study design. “Data and analysis from this study will provide a robust scientific basis for the development of prognostic models that can identify those at risk of developing HCC and other unfavourable outcomes.”
“Metabolites can be used as early indicators of health problems on the horizon. We will perform metabolic phenotyping of the cohort at high risk of developing HCC, and we should be able to identify early biomarkers that can predict the onset of HCC. The outcome could provide clues for regulating gut microbes by precision nutritional intervention to slow down or prevent the development of HCC,” said Prof Wang Yulan, Director of the Singapore Phenome Centre (SPC) at Lee Kong Chian School of Medicine, Nanyang Technological University Singapore.
Conducting the ELEGANCE study in Singapore
With 80% of HCC cases worldwide diagnosed in the Asia Pacific region, Singapore is a fitting site to conduct the study. [3] The four-year long study launched late last month will enroll 2,000 participants at risk for HCC. These include patients with liver cirrhosis, hepatitis B or C, non-alcoholic fatty liver disease (NAFLD) or non-alcoholic steatohepatitis (NASH).
The study involves public and private sector collaboration and has three tracks: 1) to evaluate the efficacy of a miRNA diagnostic kit for HCC with Singapore-headquartered multi-cancer early detection company MiRXES; 2) to develop an AI algorithm for identification of patients at-risk of developing HCC using state-of-the-art quantitative MR imaging, with digital medical technology company, Perspectum, whose Asia Pacific headquarters are in Singapore; and 3) to determine the changes in the microbiome and metabolome that lead to HCC with Southeast Asian precision gut microbiome company AMiLi.
The goal of all three tracks is early diagnosis, better and more cost effective methods for improved patient outcomes and the identification of novel therapeutic targets.
Whole-of nation effort
Healthcare institutions including National Cancer Centre Singapore (NCCS), Singapore General Hospital (SGH), National University Hospital (NUH), Changi General Hospital (CGH), Sengkang General Hospital (SKH), Tan Tock Seng Hospital (TTSH) and eight SingHealth Polyclinics (Bedok, Bukit Merah, Marine Parade, Outram, Pasir Ris, Punggol, Sengkang and Tampines) will serve as recruitment sites. Clinician-scientists and scientists from the recruiting hospitals and polyclinics, as well as academic institutions, Duke-NUS and Singapore Phenome Centre are collaborators for this study.
“There is indeed a pressing need for a collaborative multi-center study to identify patients at the highest risk of HCC in Singapore. In the era of precision medicine, we aim to develop state-of-the-art diagnostic tools to effectively diagnose HCC at an early stage. Our common goal is to reduce mortality from HCC,” said Associate Professor Jason Chang Pik Eu, Head and Senior Consultant, Department of Gastroenterology & Hepatology, SGH.
Associate Professor Dan Yock Young, Senior Consultant, Division of Gastroenterology and Hepatology, NUH said, "Liver cancer is one of the deadliest cancers in Asia. Developing new tools of screening and identification of the at risk population will allow us to detect cancer at its earlier stage, where it is potentially curative."
Patient recruitment for the ELEGANCE study
The research team is actively recruiting patients with chronic liver disease as validated by blood tests and imaging. Enrolled patients will be monitored regularly by current standard-of-care imaging and blood tests. Collection of additional clinical data, bio samples and regular follow-ups will continue at participating hospitals/clinics for up to three years after enrolment. As this is an observational research study, there will be no interventional treatment but patients diagnosed with liver cancer during the study will be treated by the respective healthcare institutions according to standard clinical practice.
For more information on the study and eligibility, please contact the study’s coordinators:
Tel: +65 6326 6573
Email:
ahcc10@nccs.com.sg
References:
[1] Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A., & Jemal, A. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 68(6), 394–424.
[2] National Registry of Diseases Office. (2021, March). Singapore Cancer Registry Annual Report 2018.
[3] National Registry of Diseases Office. (2019, November). Singapore Cancer Registry 50th Anniversary Monograph - Appendices.
© 2025 SingHealth Group. All Rights Reserved.
















































